Case Report

Low Grade Endometrial Stromal Sarcoma of Ectocervix: Report of a Case and Review of Literature.
* Reema Bhushan, Shilpi Agarwal, Sangeeta Pahuja
- *Department of Pathology, Lady Hardinge Medical College, New Delhi, India
- Submitted: Tuesday, December 27, 2016
- Accepted: Tuesday, January 31, 2017
- Published: Friday, February 3, 2017
This is an Open Access article distributed under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited
Abstract
Background
Endometrial stromal sarcomas (ESS) are rare malignant tumors of the uterus. The primary tumour can occur in extra-uterine sites like ovary, fallopian tube, cervix, vulva, vagina, omentum and retro peritoneum. Primary low grade ESS occurring in ectocervix is reported very rarely. Primary tumour in the cervix mimics a fibroid polyp and poses a diagnostic dilemma.
Case Presentation
We report a case of 43 years old perimenopausal lady who presented with bleeding
per vaginum and cervical polyp. Ultrasound revealed uterine leiomyoma in addition to the cervical polyp. Histological examination revealed epithelioid cell tumour. The diagnostic challenge was to differentiate ESS from epithelioid leiomyoma and PEcoma for which immunohistochemistry was carried out which showed positivity for
Cluster of Differentiation 10 (CD10), Estrogen Receptor (ER), Progesterone
Receptor (PR), Wilms’ Tumor 1 (WT1), Cytokeratin (CK) and Vimentin .
Conclusions
The case report emphasises the need to include ESS in the differential diagnosis of cervical polyp.
Key words
Vaginal bleeding; dysfunctional bleeding; leiomyoma; cervical polyp; epitheloid cell tumor; stromal sarcoma
Introduction
Cervical sarcomas comprise of 0.5% of all primary cervical malignancies [1]. The median age of women with low-grade ESS is between 45 and 57 years [2]. Low-grade endometrial stromal sarcoma (LG-ESS) comprises 50-60% of ESS and may arise in extra uterine sites i.e. ovary, fallopian tube, cervix, vagina, vulva, omentum, sigmoid colon, round ligament and retroperitoneum [3]. Primary low grade ESS occurring in ectocervix is very rare. After extensive research only one case report has been found [2]. This is the first case in India to be reported with ESS arising in the ectocervix. This case highlights occurrence of extra uterine ESS in the ectocervix, presenting as a cervical polyp.
Case Presentation
A 43-year-old married gravida 2 para 2 patient presented with bleeding
per vaginum of 2 months duration with a polypoidal mass coming out of vagina. Her menstrual cycle was irregular and she was on progesterone treatment for 3 months.
Per vaginal examination revealed a polypoidal growth measuring 4cm × 3 cm × 2cm arising out of the ectocervix. Ultrasound revealed endometrial thickness of 10 mm with an intramural fibroid at isthmus measuring 3 cm × 2 cm × 2 cm. A clinical diagnosis of leiomyoma uterus with cervical fibroid was considered. Her pap smear was negative for intraepithelial lesion or malignancy and her endometrial biopsy was reported as disordered proliferative endometrium. A total abdominal hysterectomy with cervical fibroid polypectomy was carried out and the specimen was sent for histopathological examination.
Gross appearance
Gross examination revealed specimen of uterus with cervix with intramural fibroid altogether measuring 14cm × 12 cm × 7 cm. Endometrial thickness vary from 5 to 10 mm. A single intramural fibroid measuring 3 cm × 2 cm × 2 cm was noted at the isthmic region. The cervical fibroid was sent separately. It measured 4 cm × 3 cm × 2 cm (Figure 1). Cut surface was grey white and glistening. No whorling, area of haemorrhage or necrosis seen.
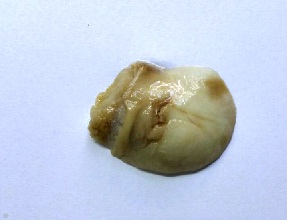
Figure 1: Cervical fibroid measuring 4 x 3 x 2 cm
Microscopic examination
Uterus showed effect of progesterone treatment. Sections from myometrium show adenomyosis with leiomyoma. Endocervix show endocervicitis with squamous metaplasia.
Sections from the cervical polyp showed diffusely infiltrating tumour tissue lined by ectocervical stratified squamous epithelium. The tumour composed of sheets of mildly pleomorphic epithelioid cells having moderate amount of eosinophilic to clear cytoplasm, round to oval nucleus with mild anisonucleosis, vesicular chromatin and mostly inconspicuous nucleoli (Figure 2).
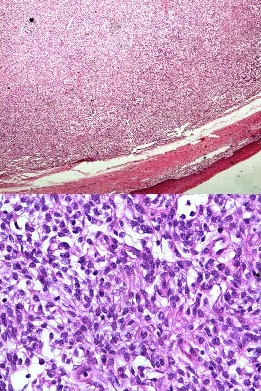
Figure 2: Photomicrograph showing (A) diffusely infiltrating tumor H&E x10
(B) showing sheets of mildly pleomorphic epitheloid cells having moderate amount
of cytoplasm with round oval nuclei H&E x40
Very few cells show small prominent nucleoli. Two mitotic figures per 10 high power fields were noted. No glandular differentiation identified. A differential diagnosis of epithelioid cells with clear cell change like epithelioid leiomyoma with clear cells, PEComa and endometrial stromal sarcoma was considered. Hence, a panel of immunohistochemical markers were put. Positive staining was noted for CD10, ER, PR, WT1, CK and Vimentin. SMA and desmin were negative (Figure 3) hence, ruling out leiomyoma. Pacoima was excluded as HMB- 45 was negative. Hence, a final diagnosis of low grade Endometrial stromal sarcoma of the ectocervix was given.
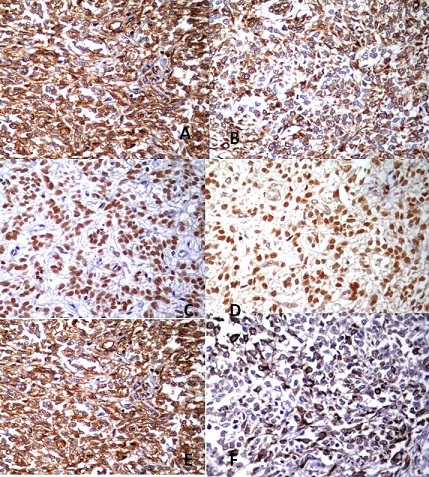
Figure 3: Immunohistochemistry showing (A) Positivity for cytokeratin, (B)
Weak immunoreactivity to Vimentin, (C) Weak positivity for Erstrogen receptor
(D) Weak positivity for progesterone receptor (E) Strong positivity for CD-10
and (F) Weak positive for WT1
Discussion
There are very few reported cases of LGESS in the cervix. Most of the cases in the literature have been described in endocervix. Boardman et al., in his study have reported a case of low grade ESS occurring in ectocervix in a 54 year old patient with carcinoma breast who was on hormonal therapy [2]. In India, two cases of ESS in cervix have been reported in 20 year and 48 year female patients [3, 4]. Both these cases have been reported in endocervix with none reported in ectocervix so far after extensive search in the literature.
The origin and biology of stromal sarcomas are not well understood. There is a relation between chromosomal aberrations and endometrial sarcomas. Chromosomal deletion on 7p was the most common finding (55.6%) in ESS [3]. The fusion of the two zinc-finger genes JAZF1 and SUZ1 at the 7p15 and 17q21 breakpoints, respectively, was described in endometrial stromal tumors by Koontz
et al., in 2001 [5]. The prevalence of the associated translocation, t(7;17)(p15;q21) in low-grade endometrial stromal sarcoma by RT-PCR [5,- 7] and FISH [8, 9] ranges from 23 to 80%.
The pathogenesis of ESS is unknown, but exposure to tamoxifen, unopposed estrogens, and conditions such as polycystic disease of ovary are implicated [10]. One fourth of patients are asymptomatic. Most patients present with abnormal vaginal bleeding, polypoidal vaginal mass and dysmenorrhoea. The other symptoms could be urinary urgency, retention of urine or constipation [3].
Primary tumour arising in the cervix mimics a fibroid polyp and poses a diagnostic dilemma. Tumors of cervix with eosinophilic to clear epithelioid cells include epithelioid leiomyoma, PEComa and ESS. Epithelioid leiomyoma show round or polygonal rather than spindle shaped cells. The nuclei are round
and relatively large. These cells have abundant clear cytoplasm and well defined cell membranes. SMA and desmin were negative in the present case, hence ruling out epithelioid leiomyoma. PEComa which shows epithelioid to spindle shaped cells with abundant clear to eosinophilic cytoplasm was also considered as a differential, but immunohistochemistry was negative for HMB 45
(Figure 4).
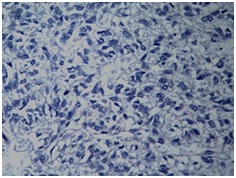
Figure 4: Negative immunohistochemistry for HMB 45
LG- ESS cells resemble proliferative phase endometrium. Cells have abundant clear to eosinophilic cytoplasm with poorly defined cell borders, round to oval nucleus, finely granular chromatin and small inconspicuous nucleoli. Positive staining was seen with CD10, ER, PR, WT1, CK and vimentin in this case. Based on histopathology and immunohistochemistry, a final diagnosis of low grade ESS was given. About one-third of patients with ESS show recurrent local disease [11, 12]. However, in present case, patient presented with the polyp for the first time. The standard surgical treatment is TAH with bilateral salpingo-oophrectomy. As the tumour is hormonally responsive, hormone replacement therapy containing estrogen and tamoxifen is contraindicated postoperatively [13].
Conclusion
We report a case of ESS which occurred at an unusual location and suggest that ESS should be included in the differential diagnosis of cervical sarcoma.
Authors' Contribution
RB: contributed to conception and design, acquisition of data
and interpretation of data, drafting the article for important intellectual
content and final approval of the version to be published.
SA: contributed to conception and design, acquisition of
data, analysis and interpretation of data, drafting the article or revising it
critically for important intellectual content and final approval of the version
to be published.
SP: contributed to conception and design, analysis and
interpretation of data, revising it critically for content, final approval of
the version to be published.
Ethical Considerations
The authors declare that the written informed consent was taken from the patient for publication of this case report. A copy of the consent is available with the authors.
Conflict of Interests
The authors declare that there are no conflicts of interests.
Funding
None declared
Acknowledgement
None
References
[1].Hasiakos D, Papakonstantinou K, Kondi-Paphiti A, Fotiou S. Low-grade endometrial stromal sarcoma of the endocervix. Report of a case and review of the literature.Eur J Gynaecol Oncol. 2007; 28(6):483-[PubMed]
[2].Boardman CH, Webb MJ, Jefferies JA. Low-Grade Endometrial Stromal Sarcoma of the Ectocervix after Therapy for Breast Cancer. Gynecologic Oncology 2000:79; 120–123.[PubMed]
[3].Jindal D, Jindal M. Primary
Endometrial Stromal Sarcoma arising from Cervix. IOSR Journal of Dental and
Medical Sciences (IOSR-JDMS) 2015(14); 12: 139-144 [Full
text]
[4].Usha M, Rau AR, Sujani B K, Uravashi T. Low grade endometrial stromal sarcoma presenting as a cervical polyp in a young female: A rare case report. Clin Cancer Investig J 2014;3:257-9 [Full
text]
[5].Koontz JI, Soreng AL, Nucci M, Kuo FC, Pauwels P, van Den Berghe H, Dal Cin P, Fletcher JA, Sklar J.. Frequent fusion of the JAZF1 and JJAZ1 genes in endometrial stromal tumours. Proc Natl Acad Sci USA2001; 98:6348–6353 .[Pubmed] [PMC]
[6].Oliva E, Leval L, Soslow RA, Herens C. High frequency of JAZF1-JJAZ1 gene fusion in endometrial stromal tumours with smooth muscle differentiation by interphase FISH detection. Am J Surg Pathol 2007; 31:1277–1284.[PubMed]
[7].Chiang S, Ali R, Melnyk N, McAlpine JN, Huntsman DG, Gilks CB, Lee CH, Oliva E. Frequency of known gene rearrangements in endometrial stromal tumours. Am J Surg Pathol 2011; 35:1364–1372 [PubMed]
[8].Hrzenjak A, Moinfar F, Tavassoli FA, Strohmeier B, Kremser ML, Zatloukal K, Denk H. JAZF1/JJAZ1 gene fusion in endometrial stromal sarcomas: molecular analysis by reverse transcriptase-polymerase chain reaction optimized for paraffin-embedded tissue. J Mol Diagn 2005; 7:388–395 [PubMed] [PMC]
[9].Huang HY, Ladanyi M, Soslow RA. Molecular detection of JAZF1-JJAZ1 gene fusion in endometrial stromal neoplasms with classic and variant histology: evidence for genetic heterogeneity. Am J Surg Pathol 2004; 28:224–232 [PubMed]
[10].Kurihara S, Oda Y, Ohishi Y, Iwasa A, Takahira T, Kaneki E, Kobayashi H, Wake N, Tsuneyoshi M. Endometrial stromal sarcomas and related high-grade sarcomas: immunohistochemical and molecular genetic study of 31 cases. Am J Surg Pathol 2008; 32:1228–1238 [PubMed]
[11].Cohen I. Endometrial pathologies associated with postmenopausal tamoxifen treatment. Gynecol Oncol.2004; 2:256–66 [PubMed]
[12].Ashraf-Ganjoei T, Behtash N, Shariat M, Mosavi A. Low grade endometrial stromal sarcoma of uterine corpus, a clinico-pathological and survey study in 14 cases. World J Surg Oncol 2006; 4:50 [PubMed] [PMC]
[13].Puliyath G, Nair MK. Endometrial stromal sarcoma: A review of the literature. Indian J Med Paediatr Oncol 2012; 33:1-6 [PubMerd] [PMC]

